WATER
Most
abundant and important biomolecule for the development of life on Earth. Water
gathers a series of unique characteristics that make it an irreplaceable
solvent in the biosphere
 Physical Properties
Physical Properties
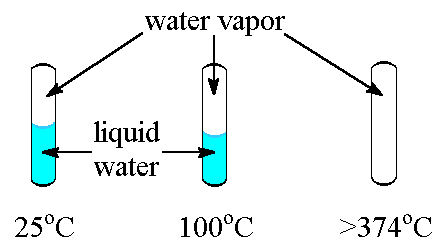
Wide range of temperature that remains in
a liquid phase. (0 – 100 °C)
Abnormal variation of density versus temperatureoSuperficial tensionoCapillarity
Its elevated dielectric constant, dipolar characteristics, specific heat and heat of evaporization.
Superfical tension
Capillarity

Abnormal variation of density versus temperatureoSuperficial tensionoCapillarity
Its elevated dielectric constant, dipolar characteristics, specific heat and heat of evaporization.
Superfical tension
Capillarity
Chemical Properties
water is a compound of hydrogen and oxygen, having the formula H2O. It is chemically active, reacting with certain metals and metal oxides to form bases, and with certain oxides of nonmetals to form acids. It reacts with certain organic compounds to form a variety of products